Scientific Commentary on a Paper about PTEN
By Mingzhen Tian
原文链接:FBXO22 degrades nuclear PTEN to promote tumorigenesis
This article recommended by Professor Guoqiang Chen is about the discovery on FBXO22 degrading nuclear PTEN for tumorigenesis. For the first time, researchers identified different regulatory mechanisms between nuclear/plasma PTEN and identified that FBXO22 promotes the ubiquitination of nuclear PTEN, which in turn degrades PTEN. This study provides a new rationale for tumor treatment strategies that target activation of PTEN.
PTEN is an important tumor suppressor gene that acts through the lipid phosphatase activity of its protein. Recent studies have shown that the phosphatase independent function of PTEN can also exert inhibitory effects on cancer, and this phosphatase non-dependent function is mediated by nuclear PTEN. Moreover, reduction of nuclear PTEN was more common than cytoplasmic PTEN in a variety of cancers. However, the regulatory mechanism of PTEN in the nucleus remains unclear. This article reports that FBXO22 can ubiquitously degrade nuclear PTEN and thus exert oncogenic effects.
First, after knocking out endogenous PTEN, the researchers found PTEN-GFP expression in the nucleus, and the expression increased after inhibiting protein degradation by MG132. In contrast, cytoplasmic PTEN was unaffected by MG132. In addition, by immunoprecipitating Ub and PTEN, they found that nuclear PTEN was more enriched in Ub than cytoplasmic PTEN and that ubiquitinated PTEN was more abundant in the nucleus after MG132 treatment. These unique findings suggest that nuclear PTEN is more sensitive to ubiquitination degradation.
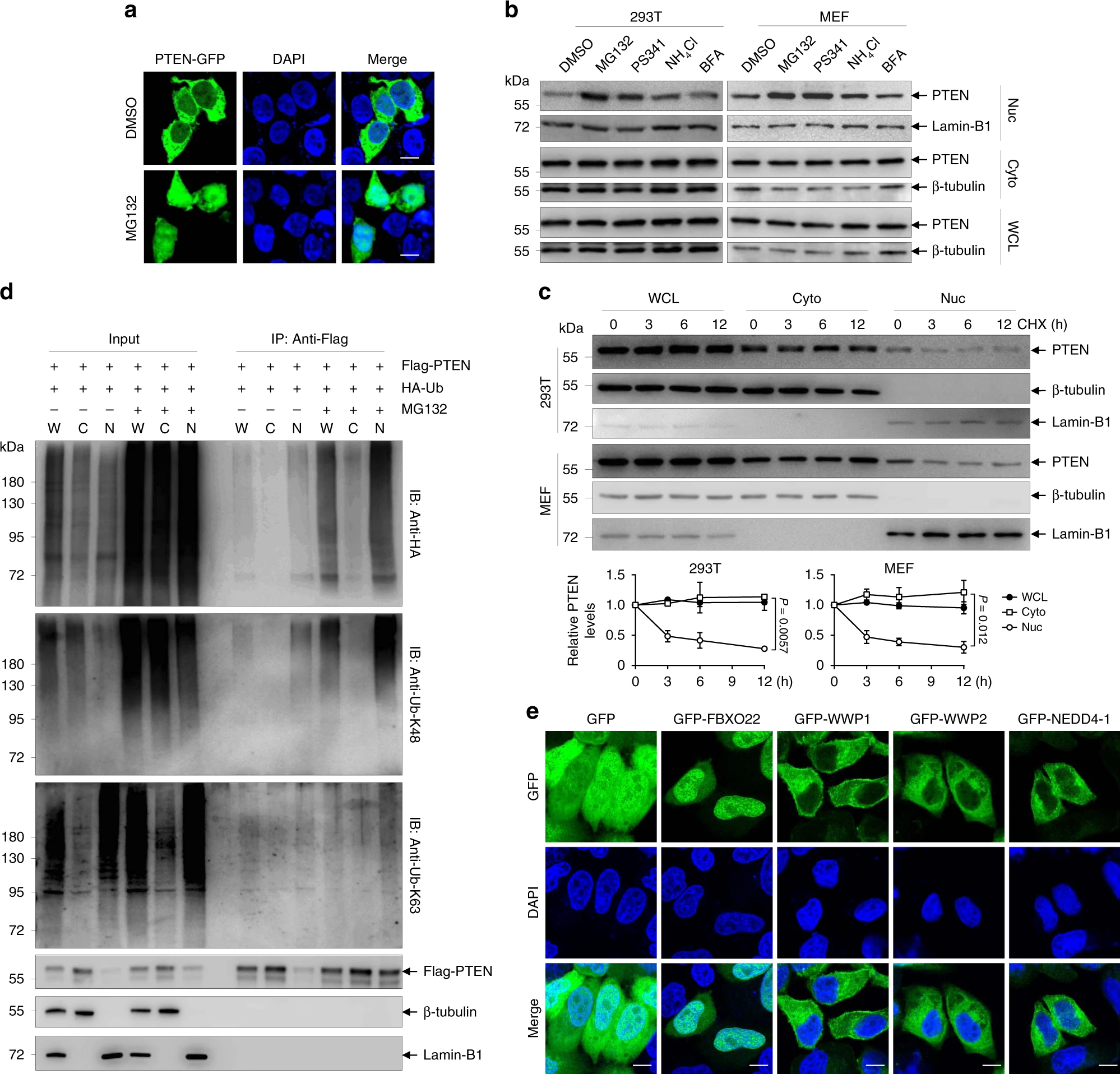
Next, the authors wanted to find the ligase for nuclear PTEN ubiquitination. They first validated the known cytoplasmic PTEN ubiquitination ligases NEDD4-1 and WWP2 and found that they did not work on nuclear PTEN. Then, they found that FBXO22 could co-localize to the nuclear PTEN. So next they started to study the interaction of FBXO22 with PTEN. They demonstrated by a series of immunoprecipitation experiments that FBXO22 is a nuclear PTEN-specific ubiquitin ligase. Further, they identified aa 366~375 of PTEN as the docking site for its binding to FBXO22.
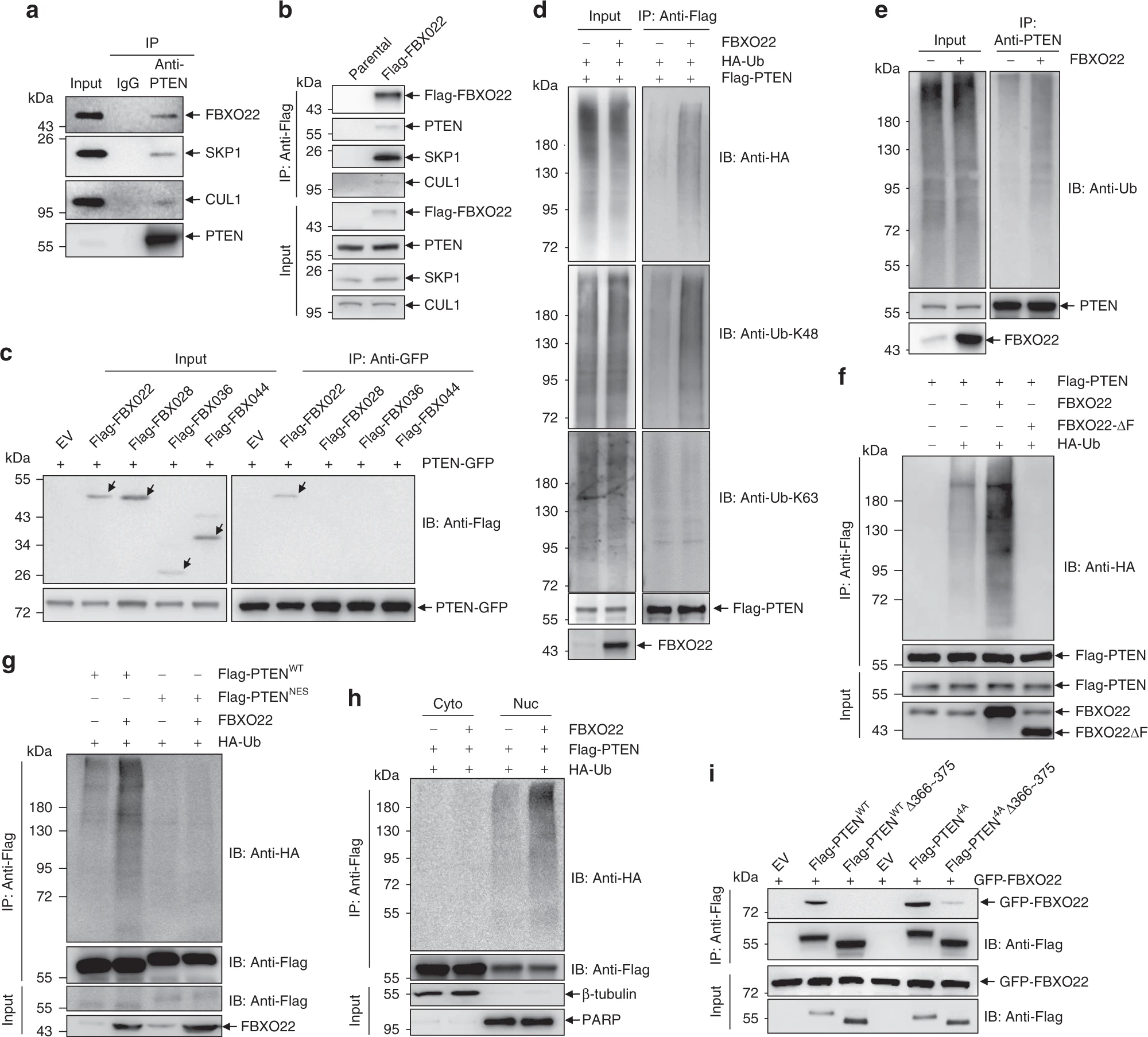
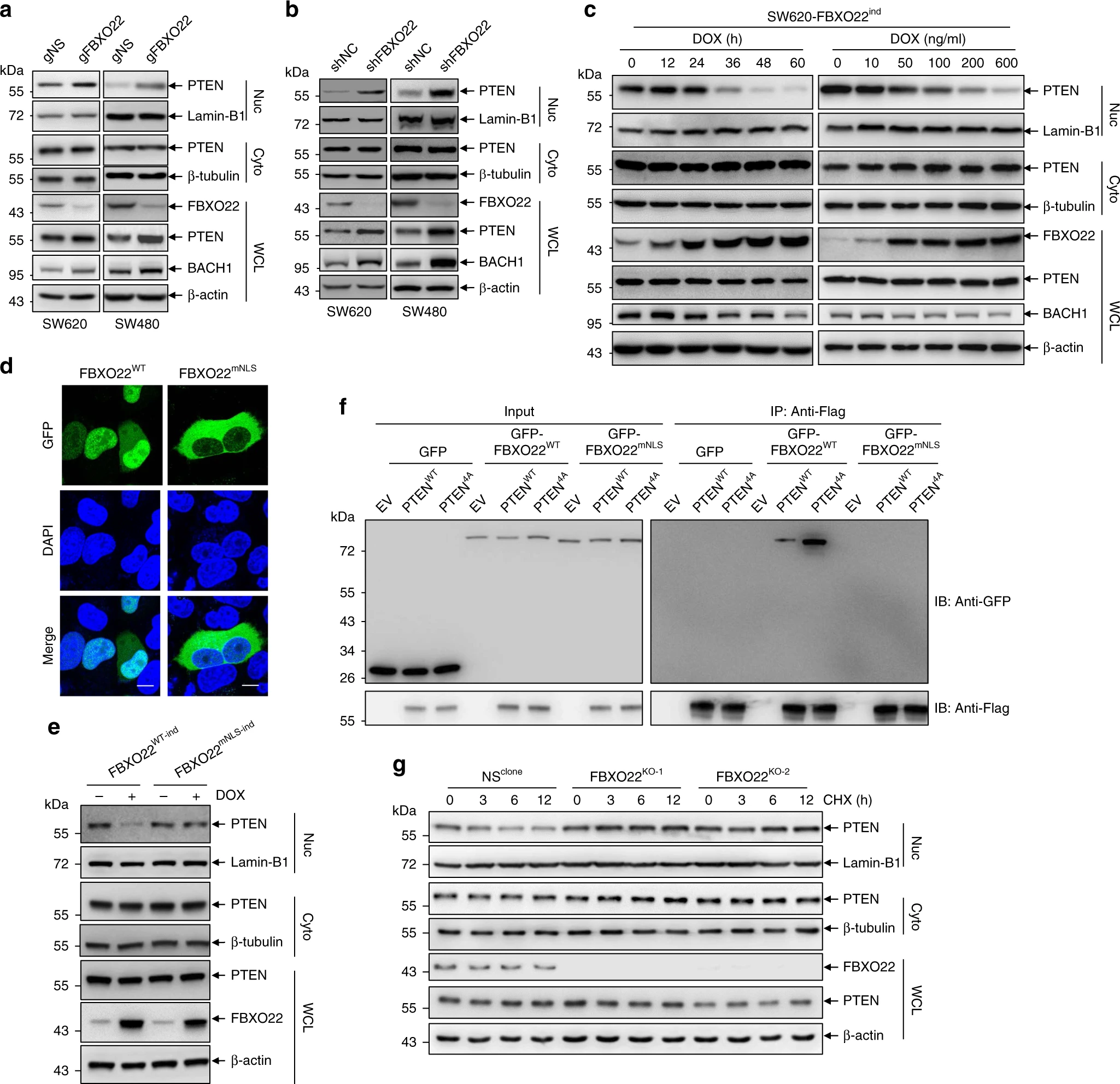
After obtaining these results, the authors went on to explore whether FBXO22 regulates nuclear PTEN in cancer. Nuclear PTEN was found to be enriched after knockdown of FBXO22 by different methods in colorectal cancer cell lines, while nuclear PTEN was reduced after induced expression of FBXO22. They also looked for a mutant of FBXO22 that could only be expressed in the cytoplasm, thus confirming that FBXO22 does not destabilizes cytoplasmic PTEN. These results demonstrate that FBXO22 can modulate the stability of nuclear PTEN.
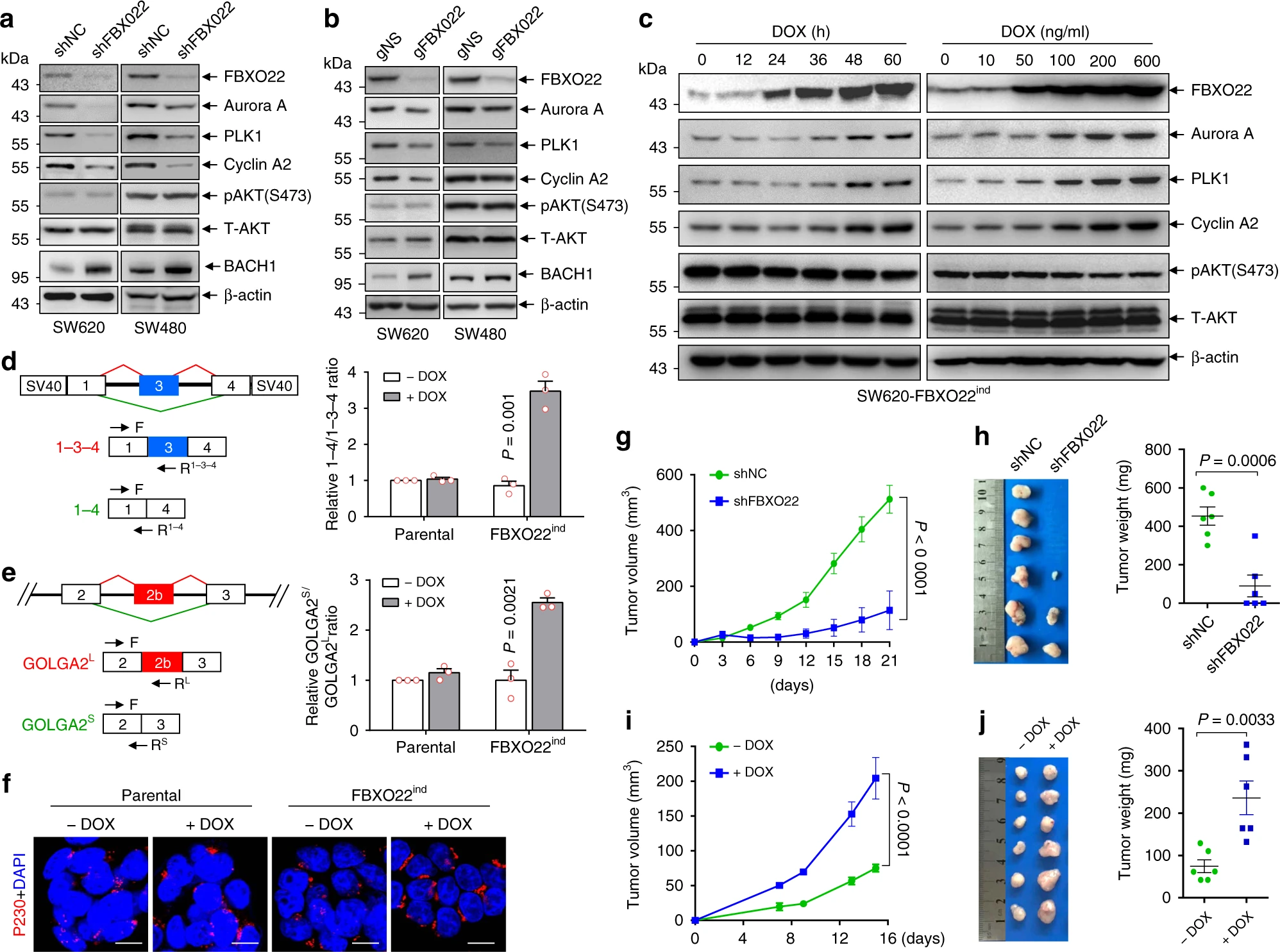
They next explored the specific mechanism by which FBXO22 regulates nuclear PTEN. First, they found that knockdown of FBXO22 promoted the phosphatase-independent function of nuclear PTEN but had no effect on the phosphatase-dependent function of cytoplasmic PTEN. Specifically, they found that FBXO22 inhibited the Golgi extension and secretion-promoting, an effect of nuclear PTEN, by interfering the alternative splicing of GOLGA2. In addition, they found that FBXO22 could induce tumor growth in mice subcutaneous tissue.
The authors then pondered whether the two roles of FBXO22, namely inhibition of nuclear PTEN downstream signaling and pro-oncogenic effects, were linked. They first searched for ubiquitination sites of PTEN by stepwise mutation strategy. After multiple rounds of screening, they identified lysine 221 (K221) as the ubiquitination site of PTEN. They then confirmed the role of its ubiquitination site by mutating this site and that FBXO22 can regulate the downstream signaling of PTEN by ubiquitinating this site.
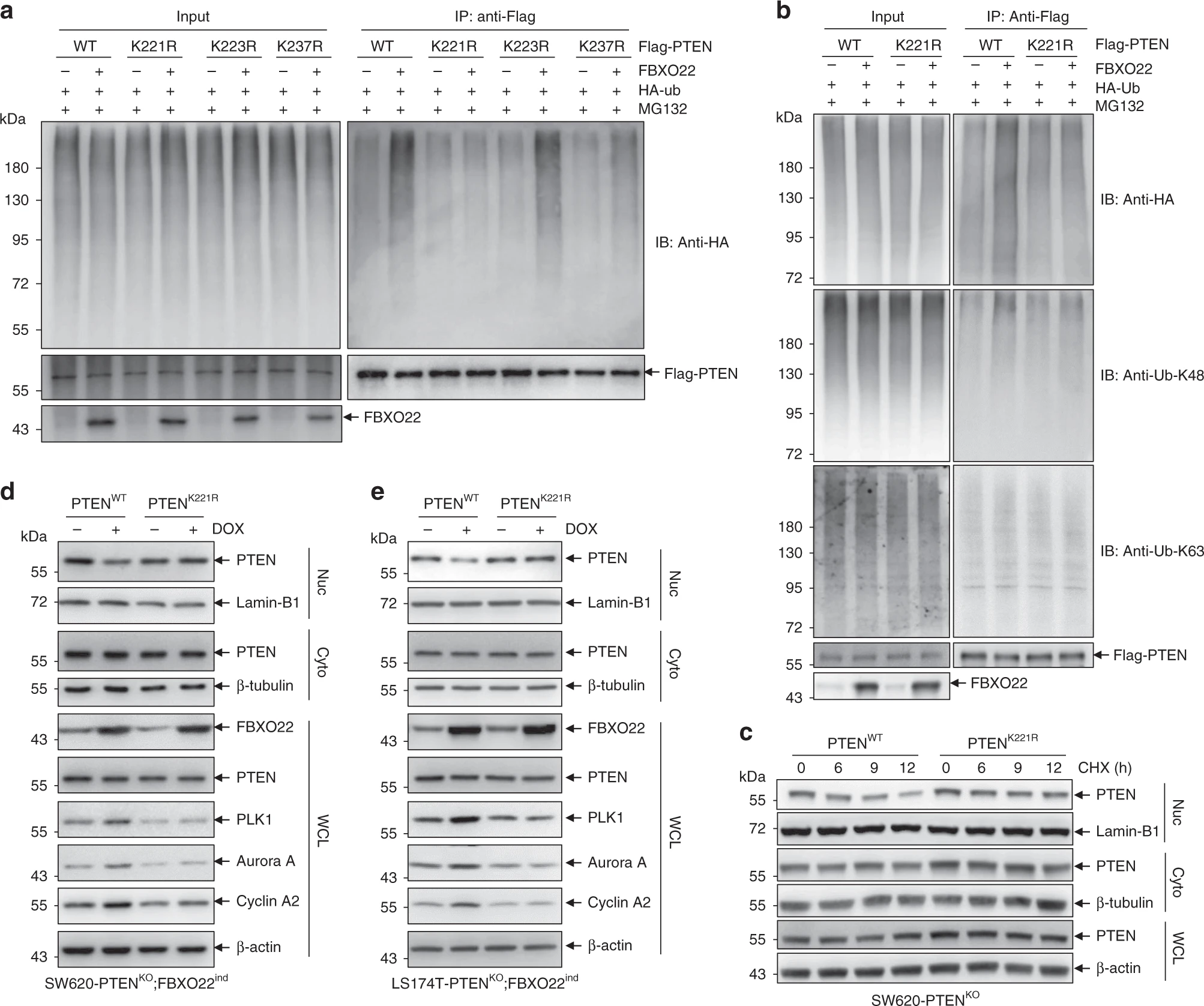
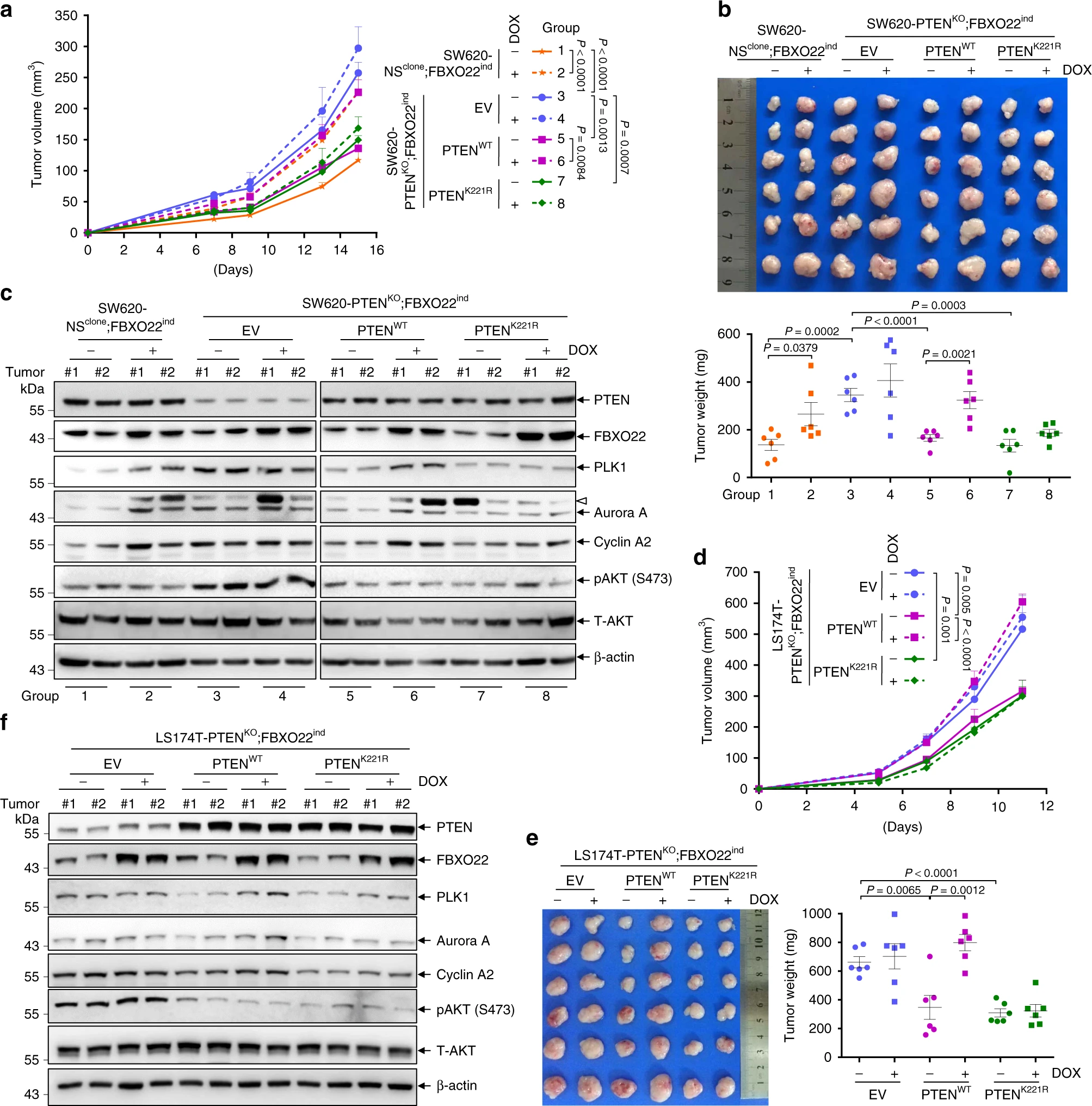
The researchers then continued to investigate the cancer-promoting effect of FBXO22 through the K221 locus. They constructed a PTEN K221 mutant mouse model and used PTEN WT as a control, and each was divided into two groups expressing and not expressing FBXO22. They found that induced expression of FBXO22 promoted cancer development, while supplementation of PTEN K221R and PTEN WT suppressed cancer. By detecting PTEN downstream signaling molecules, they found that in PTEN K221R, FBXO22 could not affect PTEN function. These results suggest that FBXO22 degrades PTEN by ubiquitinating the PTEN K221 site, which in turn promotes tumorigenesis.
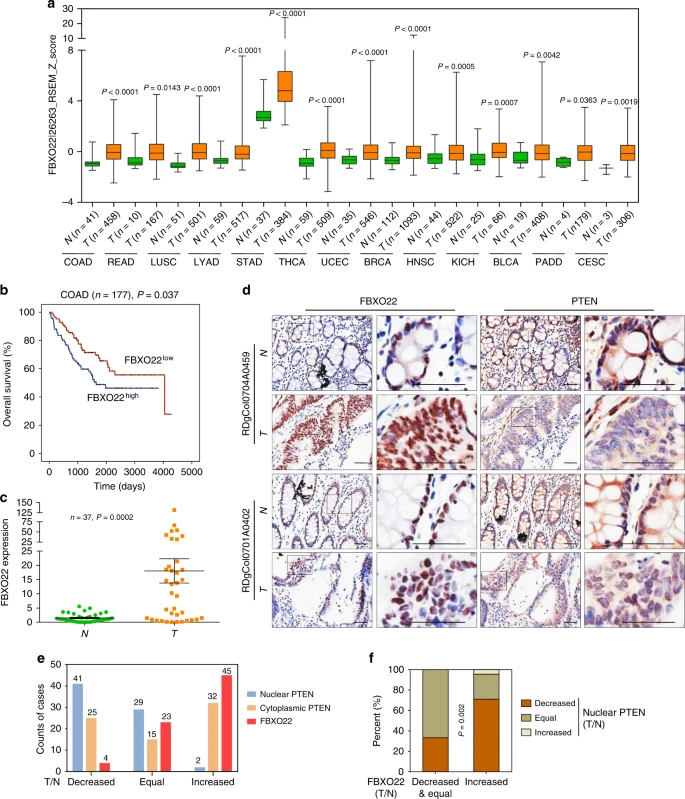
Finally, the researchers validated their findings in databases and clinical samples. First, they found FBXO22 upregulated in most cancers in the TCGA and GEO databases, especially colorectal cancer. Tissue mRNA levels in colorectal cancer patients and immunohistochemistry also supported this conclusion. In addition, nuclear PTEN expression was reduced in samples with increased FBXO22 expression, which confirms the authors’ findings.
Overall, this article identified a specific relationship between the tumor suppressor PTEN nuclear oncogenic factor FBXO22, that FBXO22 promotes tumorigenesis by degrading nuclear PTEN but not plasma PTEN through ubiquitination. This study further refines the study of PTEN function by identifying different mechanisms of cytoplasmic and nuclear PTEN. The study also identified PTEN ubiquitination sites that could be used as targets for precision cancer therapy. We can look forward to using the findings of this study to explore new approaches to tumor treatment, namely tumor suppressor reactivation.