
Scientific Commentary on a Paper about a Mechanosensitive Peri-arteriolar Niche
By Mingzhen Tian
原文链接:A mechanosensitive peri-arteriolar niche for osteogenesis and lymphopoiesis
This article recommended by Professor Dengli Hong is about the discovery of a new class of osteoblast progenitor cell. Researchers from UT Southwestern have identified for the first time adult bone marrow peri-arteriolar cells as osteogenic progenitors and have discovered mechanisms by which osteogenic progenitors are regulated by mechanical forces, partially explaining the mechanism by which exercise improves immune function and bone mineral density. Below I will briefly describe the background and specific results.
Two stem cell lineages are included in the bone marrow, hematopoietic stem cells and skeletal stem cells. They are maintained in specific niches in the hematopoietic tissue and stromal cells produce growth factors in these microenvironments. In bone marrow, leptin receptor-positive cells (LEPR+ cells) are a type of pericyte, which contains skeletal stem cells, osteogenic progenitor cells, lipogenic progenitor cells, and a variety of other cell types. LEPR+ cells are also an important source of growth factors. However, how to identify these heterogeneous LEPR+ cells remains unclear. The authors of this article have identified a class of peri-arteriolar LEPR+ cells that express osteolectin (Oln).
They first identified osteolectin expression in peri-arteriolar LEPR+ cells. They chimerized Oln with mTomato and constructed OlnmT mice to localize osteolectin. By flow cytometry they found that about 0.025% of the bone marrow cells were Oln-mTomato+ and that these cells largely expressed LEPR, accounting for 16% of all LEPR+ cells. Then by deep confocal imaging they found that Oln-mTomato+ cells were localized in agreement with peri-arteriolar LEPR+ cells. These results demonstrate that peri-arteriolar LEPR+ cells expressed osteolectin.
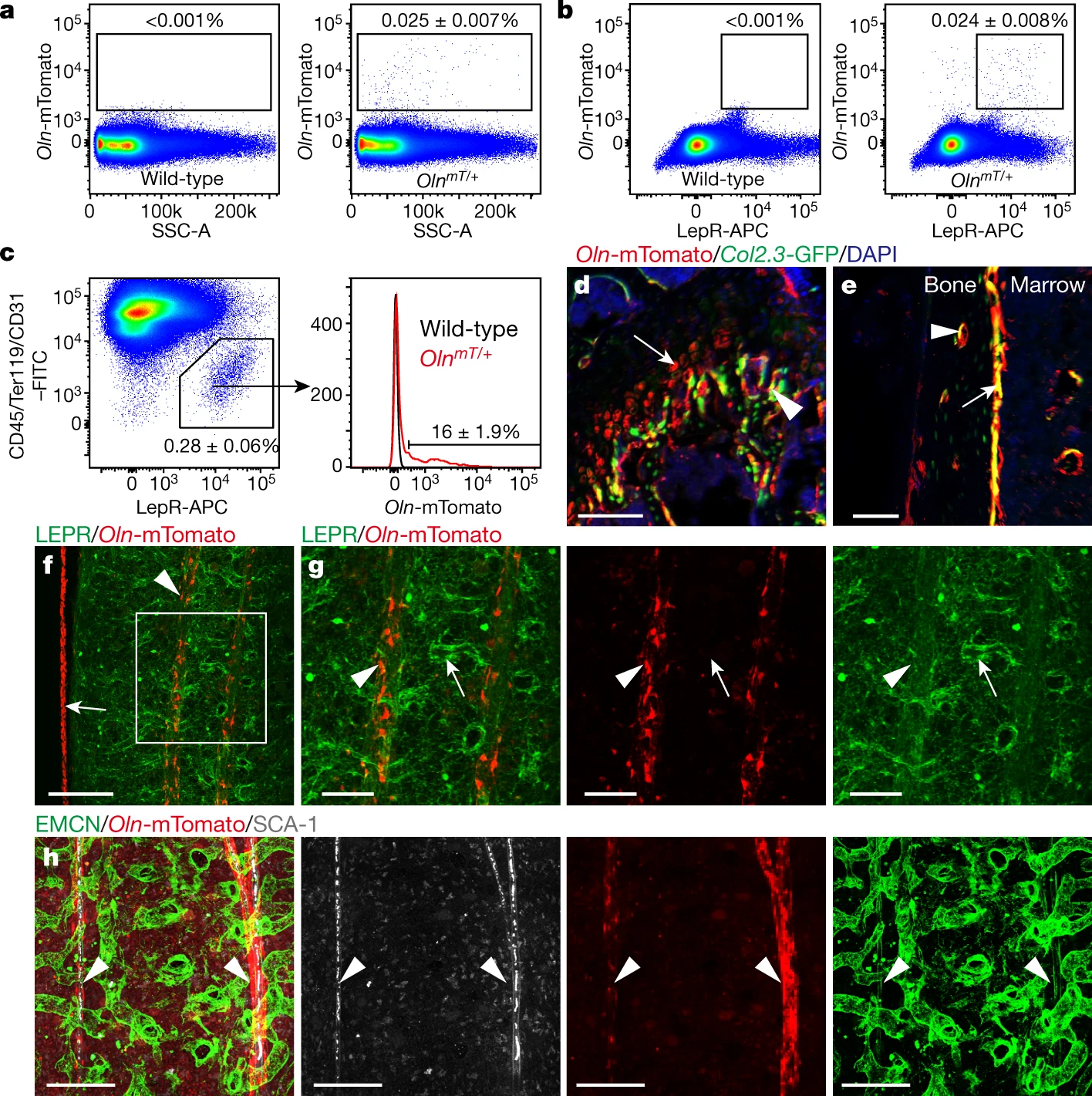
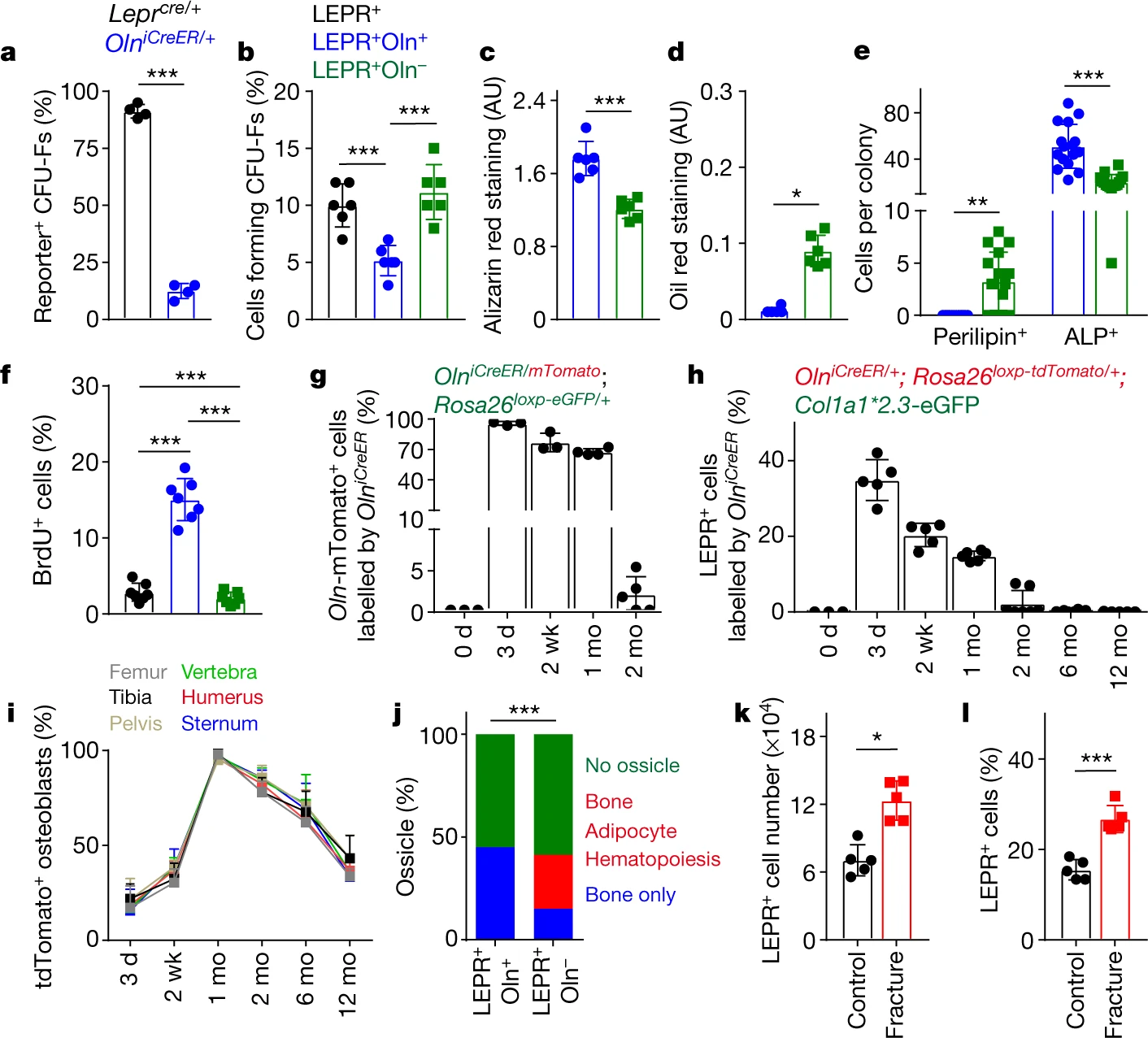
They then investigated the function of osteolectin+ cells. They found that fibroblast CFU-Fs produced by Oln+ LEPR+ cells were few. Also, when stimulated to differentiate, Oln-mTomato+ LEPR+ produced CFU-Fs developed into osteoblasts rather than adipocytes. This suggests that Oln-mTomato+ LEPR+ peri-arteriolar cells are osteogenic progenitors. Then, by examining the rate of division and survival time, the authors found that osteolectin+ cells are fast dividing cells with short survival time. The authors also found that osteolectin+ cells can continuously turnover in vivo and continue to form most of the adult neonatal skeleton.
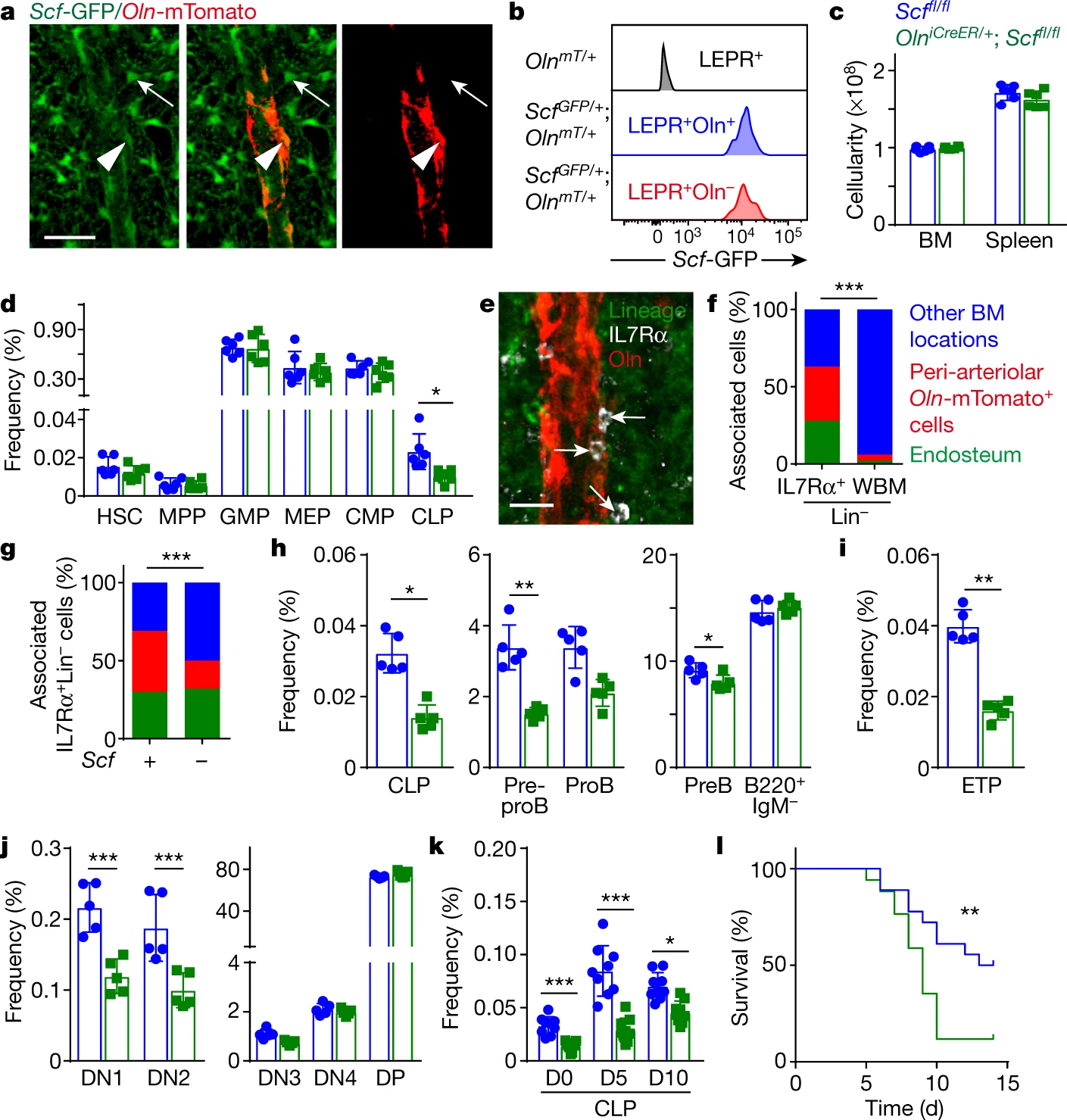
The authors then explored the effect of LEPR+ osteolectin+ cells on lymphopoiesis. They used the Cre-Loxp system to selectively knock out the Scf gene in peri-arteriolar Oln+ cells. They found that knockdown of Scf significantly reduced CLPs, while other types of progenitor cells did not change, suggesting that osteolectin+ peri-arteriolar cells may provide a niche for CLPs. They then treated the constructed mouse model with tamoxifen and after two months found that deletion of Scf reduced multiple lymphoid progenitors. By infecting Listeria monocytogenes, they also examined the effect of this niche on acute infection. They found that deletion of Scf resulted in a reduced response to acute infection. These results suggest that osteolectin+ peri-arteriolar cells provide a niche for the maintenance of lymphoid progenitors through the expression of SCF
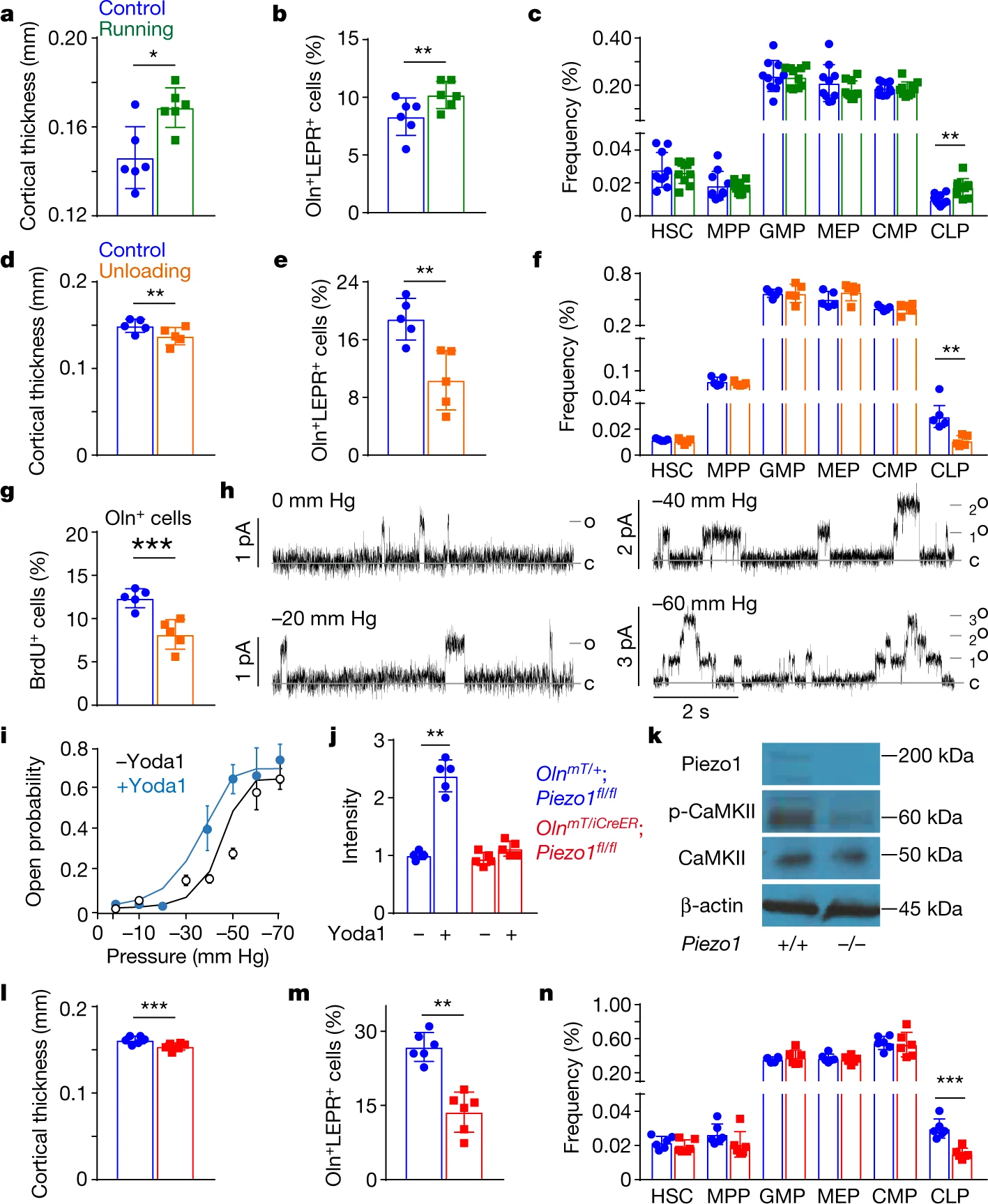
We all know that exercise improves bone quality and immunity, but the mechanism behind it is not well known. After the previous studies, we know that peri-arteriolar niches can promote osteogenesis and lymphocyte production. So, what does this novel niche have to do with exercise? The authors next explore this.
They placed a treadmill for mice to run autonomously and found that running mice had thicker bone, more Oln-mTomato+ cells and CLPs. This suggests that running promotes the function of peri-arteriolar niches. They then explored the effect of mechanical stress on osteolectin+ cells. They suspended the hind limbs of the mice to reduce the movement of the hind limbs. They found that the thickness of bone, Oln-mTomato+ cells and CLPs were significantly reduced in the lifted hind limbs. This indicates that mechanical stress is essential for the maintenance of the niche.
Then by gene expression enrichment, they found that osteolectin+ cells expressing PIEZO1. They identified piezo-like channel properties in osteolectin+ cells by a membrane clamp technique. They then knocked down Piezo1 in Oln+ cells and found that bone thickness, Oln-mTomato+ cells and CLPs were reduced. These results indicate that PIEZO1 plays an important role in maintaining the peri-arteriolar niche.
Overall, this article revealed that the mechanical forces generated by exercise can be transmitted to osteolectin+ cells in the bone marrow via the arterial vasculature. While osteolectin+ cells provide a peri-arteriolar niche expressing SCF for the maintenance of osteogenesis and lymphocyte formation.
Through the study of this article, I deeply appreciate the author’s sudden and whimsical thoughts, meticulous observations and bold attempts. The authors started from an insignificant phenotype and carried out a series of bold attempts and finally came up with excellent results. This is something I really admire. I also learned from the author’s skillful use of the Cre-Loxp system and his extensive knowledge of experimental animal models.
The weakness of this article, in my humble opinion, is that it does not explore the integration with clinical diseases. Due to the importance of this novel niche for osteogenesis and immunity, it may serve as a therapeutic target for related diseases.